#benzyne
Text
#biblically accurate benzene#seriously why does it do that#chemistry#benzyne#orgo#organic chemistry#science#pedro pascal#make your own kind of music
59 notes
·
View notes
Text
"Chcę drinka. Chcę pięćdziesiąt drinków. Chcę butelkę najczystszego, najmocniejszego, najbardziej niszczycielskiego najbardziej trującego alkoholu na Ziemi.Chcę pięćdziesiąt butelek. Chce cracku, brudnego i żółtego i wypełnionego formaldehydem. Chcę kopę metamfy w proszku, pięćset kwasów, worek grzybków, tubę kleju większą od ciężarówki, basen benzyn tak duży, żeby się w nim utopić.Chcę czegoś wszystkiego czegokolwiek jakkolwiek ile tylko się da by zapomnieć."
James Frey: "Milion małych kawałków"
#ćpun#amfetamina#narkomania#narkotyki#stymulanty#życie ćpuna#pamiętnik ćpunka#alkoholizm#i hate everything#i wanna die
40 notes
·
View notes
Text
Chcę drinka. Chcę pięćdziesiąt drinków. Chcę butelkę najczystszego, najmocniejszego, najbardziej niszczycielskiego najbardziej trującego alkoholu na Ziemi. Chcę pięćdziesiąt butelek. Chcę cracku, brudnego i żółtego i wypełnionego formaldehydem. Chcę kopę metamfy w proszku, pięćset kwasów, worek grzybków, tubę kleju większą od ciężarówki, basen benzyn tak duży, żeby się w nim utopić. Chcę czegoś wszystkiego czegokolwiek jakkolwiek ile tylko się da by zapomnieć.
#james frey#jamesfrey#frey#cytat#depressing poem#cytat po polsku#blog z cytatami#cytat dnia#cytaty#mój cytat#alkoholizm#alkohol#narkotyki#narkomania
58 notes
·
View notes
Text
harini jumaat. buat defat koko
sample tambah petroleum benzyne guna pipette 15ml each 5ml
masuk dalam shaker 2 min
pastu centrifuge 12000rpm 5 min 3 kali
pastu buang liquid supernatant
ulang lagi sekali
0 notes
Text
Пальне в серпні подорожчає: чи перевищить ціна на бензин 60 грн/л?
https://antiraid.com.ua/news/palne-v-serpni-podorozhchaie-chy-perevyshchyt-tsina-na-benzyn-60-hrn-l/
В серпні ціни на пальне зростатимуть через зростання ціни на нафту та закінчення запасів дешевого пального яке трейдери закупили до підвищення ПДВ з 7% до 20%. Разом з цим споживання палива залишається високим через жнива. Аналітик профільного видання “НафтоРинок” Олександр Сіренко каже, що все більше операторів мереж АЗС невеликими кроками наближаються в цінах бензину та […]
Повідомлення Пальне в серпні подорожчає: чи перевищить ціна на бензин 60 грн/л? з'явилися спочатку на ANTIRAID.
via ANTIRAID https://antiraid.com.ua/home/
July 31, 2023 at 11:30AM
0 notes
Photo

A star is born: Study reveals complex chemistry inside 'stellar nurseries' An international team of researchers has uncovered what might be a critical step in the chemical evolution of molecules in cosmic “stellar nurseries.” In these vast clouds of cold gas and dust in space, trillions of molecules swirl together over millions of years. The collapse of these interstellar clouds eventually gives rise to young stars and planets. Like human bodies, stellar nurseries contain a lot of organic molecules, which are made up mostly of carbon and hydrogen atoms. The group's results, published Feb. 6 in the journal Nature Astronomy, reveal how certain large organic molecules may form inside these clouds. It’s one tiny step in the eons-long chemical journey that carbon atoms undergo—forming in the hearts of dying stars, then becoming part of planets, living organisms on Earth and perhaps beyond. “In these cold molecular clouds, you’re creating the first building blocks that will, in the end, form stars and planets,” said Jordy Bouwman, research associate at the Laboratory for Atmospheric and Space Physics (LASP) and assistant professor in the Department of Chemistry at the University of Colorado Boulder. For the new study, Bouwman and his colleagues took a deep dive into one stellar nursery in particular: the Taurus Molecular Cloud (TMC-1). This region sits in the constellation Taurus and is roughly 440 light years (more than 2 quadrillion miles) from Earth. This chemically complex environment is an example of what astronomers call an “accreting starless core.” Its cloud has begun to collapse, but scientists haven’t yet detected embryonic stars emerging inside it. The team's findings hinge on a deceptively simple molecule called ortho-benzyne. Drawing on experiments on Earth and computer simulations, the researchers showed that this molecule can readily combine with others in space to form a wide range of larger organic molecules. Small building blocks, in other words, become big building blocks. And, Bouwman said, those reactions could be a sign that stellar nurseries are a lot more interesting than scientists give them credit for. “We’re only at the start of truly understanding how we go from these small building blocks to larger molecules,” he said. “I think we’ll find that this chemistry is so much more complex than we thought, even at the earliest stages of star formation.” Fateful observation Bouwman is a cosmochemist, studying a field that blends chemistry and astronomy to understand the churning chemical reactions that happen deep in space. On the surface, he said, cold molecular clouds might not seem like a hotbed of chemical activity. As their name suggests, these galactic primordial soups tend to be frigid, often hovering around -263 degrees Celsius (about -440 degrees Fahrenheit), just 10 degrees above absolute zero. Most reactions need at least a little bit of heat to get a kick-start. But cold or not, complex chemistry seems to be happening in stellar nurseries. TMC-1, in particular, contains surprising concentrations of relatively large organic molecules with names like fulvenallene and 1- and 2-ethynylcyclopentadiene. Chemists call them “five-membered ring compounds” because they each contain a ring of carbon atoms shaped like a pentagon. “Researchers kept detecting these molecules in TMC-1, but their origin was unclear,” Bouwman said. Now, he and his colleagues think they have an answer. In 2021, researchers using the Yebes 40-metre Radiotelescope in Spain found an unexpected molecule hiding in the clouds of gas of TMC-1: ortho-benzyne. Bouwman explained that this small molecule, made up of a ring of six carbon atoms with four hydrogens, is one of the extroverts of the chemistry world. It easily interacts with a number of other molecules and doesn’t require a lot of heat to do so. “There's no barrier to reaction,” Bouwman said. “That means that it has the potential to drive complex chemistry in cold environments.” Identifying the culprit To find out what kind of complex chemistry was happening in TMC-1, Bouwman and his colleagues—who hail from the United States, Germany, the Netherlands and Switzerland—turned to a technique called “photoelectron photoion coincidence spectroscopy.” The team used light generated by a giant facility called a synchrotron light source to identify the products of chemical reactions. They saw that ortho-benzyne and methyl radicals, another common constituent of molecular clouds, readily combine to form larger and more complex organic compounds. “We knew we were onto something good,” Bouwman said. The team then drew on computer models to explore the role of ortho-benzyne in a stellar nursery spread out over several light years deep in space. The results were promising: The models generated clouds of gas containing roughly the same mix of organic molecules that astronomers had observed in TMC-1 using telescopes. Ortho-benzyne, in other words, seems to be a prime candidate for driving the gas-phase organic chemistry that occurs within these stellar nurseries, Bouwman said. He added that scientists still have a lot of work to do to fully understand all the reactions happening in TMC-1. He wants to examine, for example, how organic molecules in space also pick up nitrogen atoms—key components of the DNA and amino acids of living organisms on Earth. “Our findings may just change the view on what ingredients we have in the first place to form new stars and new planets,” Bouwman said. Co-authors on the new paper include researchers at Leiden University in the Netherlands, Benedictine College in the U.S., the University of Würzburg in Germany and Paul Scherrer Institute in Switzerland.
1 note
·
View note
Text
IIT JAM Chemistry Syllabus 2022 [Updated]

IIT JAM is an examination that is conducted on a national level. The examination aims to shortlist candidates for MSc or integrated MSc programs. Institutes accept candidates into their courses through the IIT JAM examination. The test is conducted jointly by IITs and IISC. The examination is conducted by colleges that take rotational turns.
Candidates who take the IIT JAM examination can secure a seat at IISC, IITs, NITs, or CFITs. Qualifying this examination helps a student to get admission to postgraduate courses.
After completing the program successfully, candidates can get any job they aspire to in their respective specialization areas.
Students who want to pursue their post-graduation in Chemistry must be well-versed with the subject’s syllabus. Along with that, candidates must know how to prepare and not burn themselves out in the process.
In this article, we discuss IIT JAM Chemistry Syllabus. Apart from it, we discuss preparation tips as well as books that you might want for reference.
IIT JAM Chemistry Syllabus
1. Basic Mathematical Concepts
This topic includes functions; maxima and minima; integrals; ordinary differential equations; vectors and matrices; determinants; elementary statistics; and probability theory.
2. Atomic and Molecular Structure
This topic includes Fundamental particles, Bohr’s theory of hydrogen-like atoms; wave-particle duality; uncertainty principle; Schrödinger’s wave equation.
Along with the ones mentioned, you also have quantum numbers; shapes of orbitals; exclusion principle of Hund’s rule and Pauli’s; details of the electronic configuration of simple homonuclear diatomic molecules, etc.
3. Theory of Gases
The topic includes the following- Equation of state for ideal and nonideal gases; Kinetic theory of gases; distribution law of Maxwell-Boltzmann and concept of equipartition of energy.
4. Solid State
The topic includes the following- Crystals and crystal systems; X-rays; the structures of NaCl and KCl; close packing; the concept of atomic and ionic radii; radius ratio rules as well as lattice energy; Born-Haber cycle; isomorphism and heat capacity of solids.
5. Chemical Thermodynamics
The topic includes the following subtopics- Reversible and irreversible processes, the concept of first law and its application to ideal and nonideal gases, thermochemistry, the second law is in detail, Entropy and free energy, and criteria for spontaneity.
6. Chemical and Phase Equilibria
The topic includes the following- Law of mass action; Kp, Kc, Kx and Kn; effect of temperature on K; ionic equilibria in solutions; pH and buffer solutions to hydrolysis; solubility product; phase equilibria–phase rule.
Also, its application to one-component and two-component systems; colligative properties, etc. all are included here.
IIT JAM Organic Chemistry Syllabus
1. Basic concept and Stereochemistry
This topic includes electronic effects that cover resonance, inductive, and hyperconjugation. Apart from the mentioned ones, you have got optical isomerism in compounds with and without any stereocenters such as allenes, biphenyls, etc.; the conformation of acyclic systems includes substituted ethane or n-propane or n-butane as well as cyclic systems containing mono- and di-substituted cyclohexanes, etc.
2. Organic Reaction Mechanism and Synthetic Applications
This topic includes the following- Chemistry of reactive intermediates covering carbocations, carbanions, free radicals, carbenes, nitrenes, benzynes, etc.; Hofmann-Curtius-Lossen rearrangement Wolff rearrangement, Simmons-Smith reaction.
It also includes Reimer-Tiemann reaction, Michael reaction, Darzens reaction, Wittig reaction, and McMurry reaction; Pinacol-pinacolone, Favorskii, benzilic acid rearrangement, dienone-phenol rearrangement, Baeyer-Villeger reaction, and many more.
3. Qualitative Organic Analysis
This topic includes the following- Different functional groups by chemical tests along with elementary UV, IR, and 1H NMR spectroscopic techniques as tools for structural elucidation.
Syllabus for IIT JAM Inorganic Chemistry
1. The Periodic Table
This topic includes the following- Element’s periodic classification and periodicity in all the properties along with general methods of isolation and the process of purification of elements. Everything related to the periodic table is essential. You must remember all the elements by heart.
2. Chemical Bonding and Shapes of Compounds
This topic includes different types of bonding, details of VSEPR theory and shapes of molecules, hybridization, and dipole moment. It also defines ionic solids and the structure of NaCl and CsCl, diamond and graphite, lattice energy, etc.
3. Main Group Elements, especially for s and p blocks
This topic includes the following- Basic concepts on group relationships and gradation in properties; formation of electron-deficient compounds containing some elements from the primary group.
Some preparation tips for IIT JAM
Before you start preparing for your IIT JAM Chemistry paper, why don’t you go through some of the preparation tips?
1. You can start with the syllabus and exam pattern
Before beginning with the IIT JAM Chemistry preparation, you should know the complete syllabus. Knowing all the topics will help you to plan accordingly. The more clarity you have about the IIT JAM Chemistry Syllabus, the more precise you will be about proceeding with the preparation plan
2. Go through the previous year question papers
To have a grasp on your content, practicing questions is very important. So you must refer to the previous year’s question papers. Doing a lot of it can help you understand the question pattern, assess the difficulty levels, and the like.
Moreover, you will be able to identify both your weak and strong points. And this, in turn, will help you to improve a lot!
You can sign up for any coaching center that provides good guidance. It is a good idea to join some coaching institutes.
These institutes make sure to keep you in a proper drill. Such coaching centers are excellent, from conducting regular tests to updating students with all the latest news and changes.
These mock tests are helpful as you can evaluate yourself with respect to others as well as understand where you stand.
3. While studying, make sure to make notes for later reference
Making notes while you study is always recommended by teachers. You make notes that can come in handy for later use. If it is in your own language you can understand better. Also, you can highlight certain important portions which can be useful for later reference.
4. Do not jump from one chapter to the other without finishing the first one. Be systematic
You must have a systematic approach to your preparation. Make sure to complete a topic before going on to the next. Be sure what you want to start with. It is important that you plan and proceed.
5. Revise before you move on to the next the topic
Revision plays quite an important part in preparing for the IIT JAM examination. Once you are done with a topic or a chapter, revise it or practice active recall.
It is necessary to boost your confidence as well as to test your preparation and memory.
6. Last but not least, do not forget to take some time off and rest one a while. Remember, health is wealth!
Make sure you take adequate rest. You do need to have dedicated preparation for your entrance examination, but you must take some time off. Otherwise, you might have burn-out symptoms.
You might feel anxious, lethargic, nervous, on the edge, irritated, etc. And all these are likely to affect your health as well as your academics. So, it is better that you take good care of both your mental and physical health.
0 notes
Text
Negative Review: Elanyx, #453014
Who: Elanyx, #453014
What: Sales, auctions, etc.
Why: Scamming users out of special ID dragons, general shady behavior
When: This has been going on for a while, so it’s impossible to pinpoint where exactly it started.
Explain:
Elanyx, formerly known as Silverwolv, has been scamming users out of dragons with special IDs and reselling them for extreme profits. Normally this wouldn’t be an issue, but the method that they use is very scummy and shady.
(x)
Here she is reselling that same dragon. It was immediately autobought for 400kt/g.

(x)

(x)
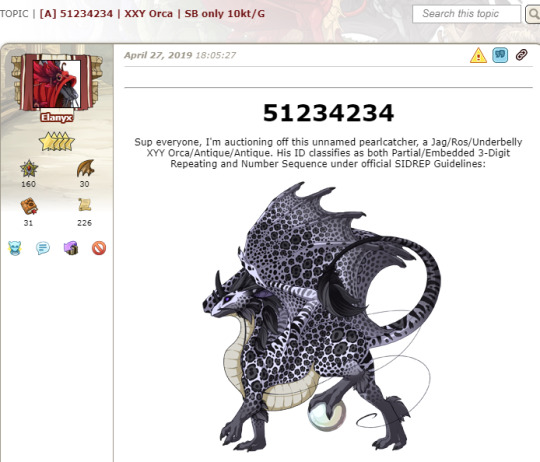
(x) (Also note that she claims it XXY first, then XYY.)

(x)
Reselling those dragons again:
(x)
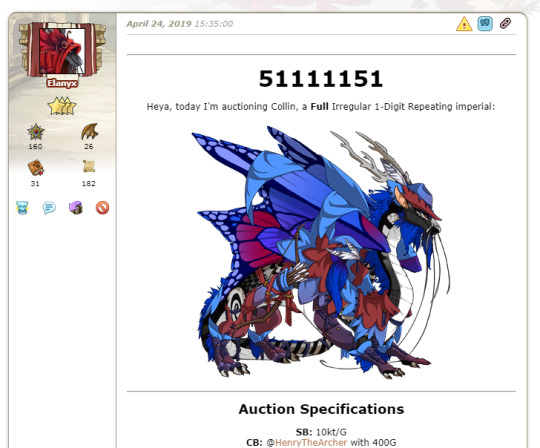
(x)

(x) They attempted to get another dragon but the owner was informed that the dragon was worth something.
They’ve admitted to this method on the old SIDREP thread before it was removed.

(x)
They’re also encouraging another user to do the same things they’re doing.
(x)
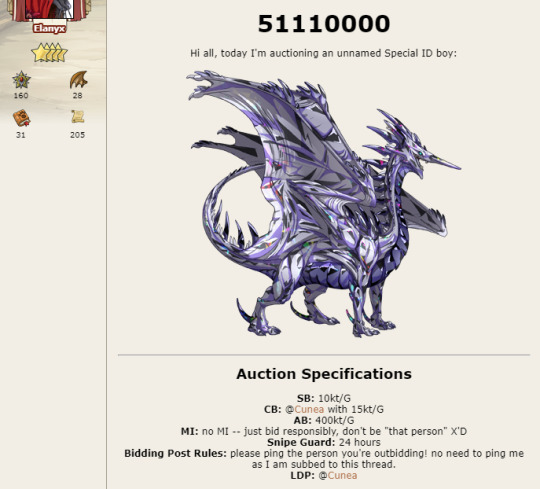
They also attempted to end an auction early. They claimed that they misread their post, but given their current behavior I wouldn’t doubt it if they truly did want to end it early. The auction was for #51110000, screenshot of said auction above.

(x)
Note that my bid was at 15:35:22 server time. Here you can see they acknowledged my bid.
(x)
Cunea’s bid was at 22:28:47.

(x)
I was given this ping at 10:52:42 on April 25th. There was still approx. 12 hours left in the auction.

They also knowingly lowball users for special ID dragons.

(x)
There’s also some proof floating around that she’s using the guise of a boyfriend and sibling to get away with multiaccounting, but that in itself is an entirely different can of worms and can be read about here. (Archive)
115 notes
·
View notes
Note
hi there i am bored! your dragons are extremely pretty :>
hi bored im net
5 notes
·
View notes
Text
nie mg sie doczekac az zrobie sb prawko i bede jezdzila po miescie puszczajac stjarnflocka
#pale diesel leje benzyne benzyneeee#jeszcze jak bdz mnie stac na benzyne to bdz supi#anyways stream audiotele bo szafker snapped mowie wam.#online and a real bastard about it
2 notes
·
View notes
Text
Ціни на бензин після підвищення податків: що змінилося?
https://antiraid.com.ua/news/tsiny-na-benzyn-pislia-pidvyshchennia-podatkiv-shcho-zminylosia/
Із 1 липня почали діяти довоєнні податки на пальне. Очікується, що податкове навантаження на бензин призведе до його подорожчання на 11 грн. Як вже змінились ціни і чому подорожчання не таке стрімке, як прогнозувалося раніше, розповів директор Консалтингової групи А-95 Сергій Куюн. Про це повідомляє АНТИРЕЙД з посиланням на «МiнфiнМедiа». Динаміка цін Після повернення повної […]
Повідомлення Ціни на бензин після підвищення податків: що змінилося? з'явилися спочатку на ANTIRAID.
via ANTIRAID https://antiraid.com.ua/home/
July 19, 2023 at 09:00AM
0 notes
Text
Chcę drinka. Chcę pięćdziesiąt drinków. Chcę butelkę najczystszego, najmocniejszego, najbardziej niszczycielskiego najbardziej trującego alkoholu na Ziemi.Chcę pięćdziesiąt butelek. Chce cracku, brudnego i żółtego i wypełnionego formaldehydem. Chcę kopę metamfy w proszku, pięćset kwasów, worek grzybków, tubę kleju większą od ciężarówki, basen benzyn tak duży, żeby się w nim utopić.Chcę czegoś wszystkiego czegokolwiek jakkolwiek ile tylko się da by zapomnieć.
12 notes
·
View notes
Photo

[Reactions of potassium amide
With halobenzenes in ammonia
Via benzyne intermediates occur
Bergstrom and associates did report,
Based on two-component competition runs,
Bromobenzene the fastest to react,
By iodobenzene closely followed,
The chloro compound lagging far behind,
And fluorobenzene to be quite inert
At reflux (-33°).
Reactions with para-dihalobenzenes,
In which the halogens were not the same,
The same order of mobility revealed,
But differences in reactivity
Were somewhat less in magnitude.
The irregular mobility rank
Explanation finds in the mechanism
Whereby arynes are formed. There are two steps:
Abstraction of the ortho proton
And the expulsion of the halogen
From the anion intermediate.
In Scheme I the mechanism is set forth.
Here proton removal is favored, in rate
And in respect to equilibrium,
By high electronegativity
Of halogen. But the expulsion step
Is faster in the opposite order.
According to the evidence, for both
Iodine and bromine step 1 limits rate.
But on the other hand, the setting free
Of halogen determines total rate
For chlorine and fluorine atoms on the ring.
We have repeated the experiments
With dihalobenzenes of Bergstrom’s group.
They are extended to the isomers
Meta and ortho, and to the action
Of potassium anilide reagent.
Throughout, halide ions have been determined
By potentiometric titration
In which end points for diverse halide ions
Are discrete, and easy to recognize.
Nitrogenous products were not assayed.
Results
Data for reactions of all nine mixed
Dihalobenzenes (excluding fluorine)
With four equivalents of amide base
Are set forth in Table I. Reactions
With the same base in deficiency
Appear, for six substrates, in Table II.
In Table I, more than one halide ion
Is set free from each dihalobenzene
Molecule. This suggests the possibility
That maybe haloanilines too react
With potassium amide. In Table III [...]]
We also note footnote 2, which reads:
[Note from Editor—Although we are open to new styles and formats for scientific publication, we must admit to surprise upon receiving this paper. However, we find the paper to be novel in its chemistry, and readable in its verse. Because of the somewhat increased space requirements and possible difficulty to some of our nonpoetically inclined readers, manuscripts in this format face an uncertain future in this office. However, we take this opportunity to encourage readers and authors to examine carefully a new format represented by the articles on pages 3591–3646 and the Editor’s Notice in the November 1970 issue of this journal.]
Bunnett and Kearley (1971)
213 notes
·
View notes
Text
🎵 The 5:15 from Duluth
Oh my it just derailed
The toxic waste is spillin'
The conductor's been impaled
A Benzyne cloud has risen,
And the whole town's startin' to cough
Cough cough, cough cough
Within a matter of daaaaays
All of our skin will fall off 🎵
6 notes
·
View notes