#hydrogen bonding
Text
Alcohols are polar compounds with the oxygen atom bearing a partial negative charge and both the carbon atom and hydrogen atom bonded to it bearing partial positive charges.
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quote#chemistry#nonfiction#textbook#alcohol#polarity#oxygen#negative charge#positive charge#hydrogen bonding#chemical bonding
4 notes
·
View notes
Text
Alcohols have higher boiling points than alkanes with the same or a similar number of electrons, because alcohols are polar and can associate in the liquid state by hydrogen bonding as depicted for ethanol in figure 19.4.
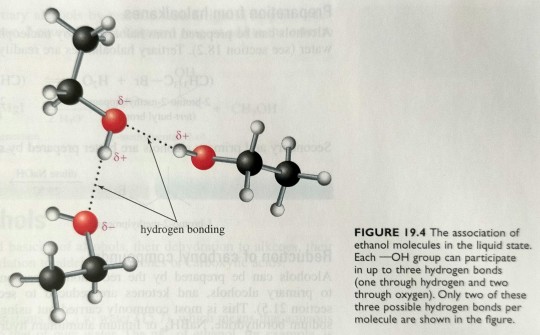
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#ethanol#alcohol#boiling point#alkane#hydrogen bonding#liquid#chemical reactions
3 notes
·
View notes
Link
Ever wondered how science captures the tiniest moments in time? Dive into the fascinating world of attosecond spectroscopy with our latest article! Discover how scientists are unveiling electron dynamics and opening new frontiers in physics and chemistry. #AttosecondSpectroscopy #ScienceRevolution
0 notes
Note
Is the REAL @swedenish* free yet??
Hmm hmm lets check
Nope.. 😔😔
I should start a blog posting every day like “is swedenish username free yet?” The answers gonna be no until I die, methinks 😣
#I check every time I’m reminded#the h will hang off like a hydrogen bond off a complex molecule foreva#at this point enough people tag this username it’s prolly not a good idea to change it BAHA 😭#asks#veradragonjedi
21 notes
·
View notes
Text
The assignment was due days ago so I was gonna take the F (sorry Kendall! it’s the anxiety of being late 😔) but Monty tagged me <3 and I’d do anything for Monty

i don’t know who’s already done this but i’m tagging friends <3 template is below :) @by-moonflower @theloveinc @boosyboo9206 @osaurimiya @imlevis @madisnorm @tteokdoroki

#reinas.inretrograde#i wanted to do one for osamu but these take thought! time! effort!#just know he is loml. my life partner. my work partner. my lover. my headache. we are hydrogen bonded. FOR LIFE#comm was done by amefurinn!
26 notes
·
View notes
Text
it's 12:30 am and i am thinking about water smiling like an idiot it's charged but only slightly isn't that silly and the ball and stick model with out the sticks looks really funny and surface tension and big scary ocean with flapping animals in it and I water cooking so cool so
#vee chats#not atsv related#isn't it so silly how it's called hydrogen bonding but it's not actually a type of bonding it's an intermolecular force#did you know dissolving is actually just the ions in a compound becoming attracted to the different charges in water and separating#guys civilian firearm possession may be illegal in the UK but
7 notes
·
View notes
Text

release — an unattainable dream
#furina#genshin impact#doodelles#unfortunately the hydrogen bonds in her are too strong and she cannot be hydrolysed
14 notes
·
View notes
Text
how hard is it to find a pretty boy who wants to talk about footballers kissing and ionic bonding????
#the two halves of my personality#also isn't 30 cuz that's a Problem™#also hey did u guys know water has an uncharacteristically high boiling and melting point?#its because the abundance of hydrogen molecules leads to a high amount of hydrogen bonding between them!#daniel.txt
7 notes
·
View notes
Text
Amines are polar compounds, and primary and secondary amines associate by intermolecular hydrogen bonding.
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
0 notes
Link
Perovskite solar cells (PVSCs) are a promising alternative to traditional silicon-based solar cells because of their high power-conversion efficiency and low cost. However, one of the major challenges in their development has been achieving long-term stability. Recently, a research team from City University of Hong Kong (CityU) made a breakthrough by developing an innovative multifunctional and non-volatile additive which can improve the efficiency and stability of perovskite solar cells by modulating perovskite film growth. This simple and effective strategy has great potential for facilitating the commercialisation of PVSCs.
"This type of multifunctional additive can be generally used to make different perovskite compositions for fabricating highly efficient and stable perovskite solar cells. The high-quality perovskite films will enable the upscaling of large-area solar panels," explained Professor Alex Jen Kwan-yue, Lee Shau Kee Chair Professor of Materials Science and Director of the Hong Kong Institute for Clean Energy at CityU, who led the study.
PVSCs have attracted significant attention due to their impressive solar power conversion efficiency (PCE). Since perovskites can be deposited from solutions onto the fabrication surfaces, PVSCs have the potential to be applied in building-integrated photovoltaics (BIPV), wearable devices, and solar farm applications. However, the efficiency and stability are still affected by the severe energy loss associated with defects embedded at the interfaces and grain boundaries of the perovskites. Therefore, the intrinsic quality of perovskite film plays a critical role in determining the achievable efficiency and stability of PVSCs.
Read more.
#Materials Science#Science#Perovskites#Solar power#Materials processing#Molecules#Hydrogen#Hydrogen bonding
7 notes
·
View notes
Text
This is illustrated in figure 16.16, which shows the uniform distribution of the outer bonding electrons in pentane.

"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#alkane#pentane#hydrocarbons#hydrogen#carbon#electron density#polarity#nonpolar#chemical bonding
9 notes
·
View notes
Text
why’s she thick 👀
23 notes
·
View notes
Text
Types of bonding: ionic, covalent, metallic, hydrogen, and trauma
12 notes
·
View notes
Note
to me, you’re hydrogen because you are my number 1 - ♦️
if im hydrogen then you must be oxygen, nitrogen, or flourine, because our bond is super strong ^^
6 notes
·
View notes
Text
science is an exercise in gratitude, i think
#ramblings#i'm basically unable to feel gratitude without guilt for having a good life#but the truths of the universe. the very nature of the world we live in. there's no possibility for injustice in that#no one can take away the perfect alignment and sizing of the moon between us and the sun that lets us have TWO kinds of eclipse#no one is denied the beautiful strangeness of water and hydrogen bonding that lets life as we know and experience it exist#the scattering of light in air that grants us pretty blue skies and fiery sunsets cannot be unfairly given or taken#me when i am sentimental over physics#it's a million miracles stacked on top of each other. what offering from the void could usurp the gift of life already given#i need to lie down
6 notes
·
View notes
Text
Went to a workshop on how to write the literature rescue for my thesis and now my stress levels are through the roof. I already feel so behind.
We had a useless year phd student who has done, as far as I can tell, absolutely nothing for the last two years while also constantly increasing my workload by skipping out on her own admin commitments and dealing group meetings. As a result I’ve had to spend the last two months (where I should have been at home recording from covid) at work doing including and training for four new masters students and one new PhD student because it turns out she literally does not know anything about our field. She could not show an undergrad first year how to use our software to do an entry level task. She cannot answer questions like “what are you studying and how are you studying it?”
I’m very tired and I feel extremely behind because I’ve been doing this shit instead of my PhD, and I’m only just now starting to figure out exactly how much I should have already been doing.
I hate her and I’m so fucking glad she quit. Enjoy failing your second year milestone because you decided to spend two years fucking around and ignoring every single piece of actionable feedback we gave you.
#PhD life#Seriously it’s been two years and she still hasn’t learned high school chemistry#WE ARE A CHEMISTRY DEPARTMENT#TWO OF US ARE ACTUAL FOR REALS CHEMISTS.#we gave her resources. We offered her training. we told her that she absolutely needed to know these things#And this bitch still hasn’t bothered to learn what a hydrogen bond is#anyway turns out she also lied about [redacted] so if she doesn’t just quit she’s probably going to get dismissed for academic misconduct#before she even has time to flunk out#idk#phd’s are hard enough without adding in someone who literally doesn’t do *anything*
5 notes
·
View notes